Description
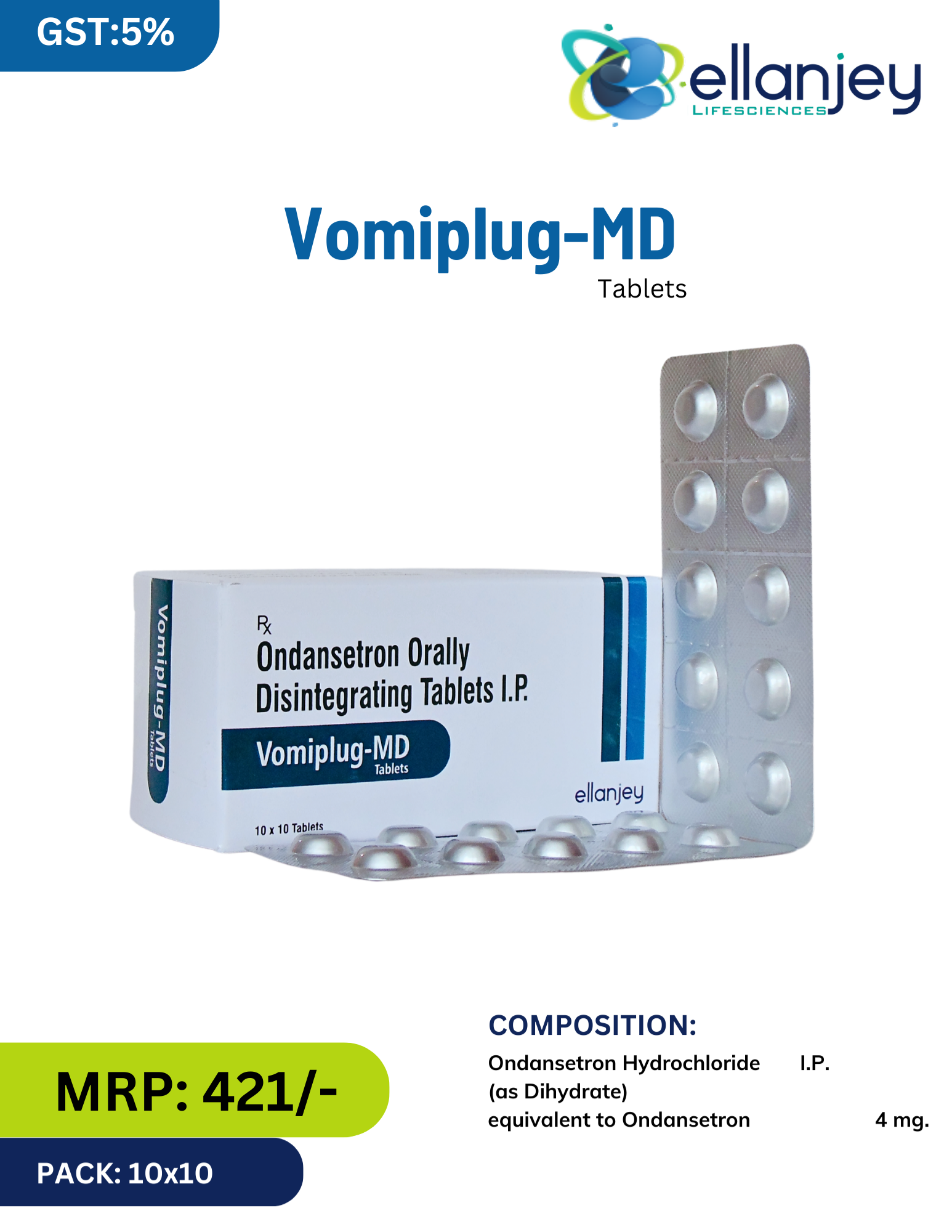
Vomiplug-MD by Ellanjey is a potent, fast-acting antiemetic medication containing Ondansetron Hydrochloride (as Dihydrate) equivalent to Ondansetron 4 mg in a convenient orally disintegrating tablet (MD – Mouth Dissolving) formulation. Ondansetron is a highly selective serotonin 5-HT₃ receptor antagonist that is widely regarded as a first-line agent for the prevention and treatment of nausea and vomiting induced by chemotherapy, radiotherapy, and surgery.
Key Benefits:
-
Powerful antiemetic – Highly effective for chemotherapy, radiation & post-surgery nausea/vomiting
-
Selective 5-HT₃ antagonist – No extrapyramidal symptoms (unlike metoclopramide)
-
Orally disintegrating (MD) formulation – Dissolves on tongue in seconds; no water needed
-
Fast-acting – Rapid buccal absorption leads to quicker onset than conventional tablets
-
Ideal for nauseous patients – Perfect for those who cannot swallow whole tablets
FAQs :
1. What is Vomiplug-MD used for?
Vomiplug-MD is used to prevent and treat nausea and vomiting caused by chemotherapy, radiation therapy, surgery (postoperative nausea and vomiting), and severe gastroenteritis. It is also used for hyperemesis gravidarum (severe morning sickness) in pregnancy.
2. How do I take Vomiplug-MD orally disintegrating tablets?
Peel the foil to remove the tablet (do not push through). Place the tablet on your tongue – it will dissolve within seconds. No water is needed. For best results against chemotherapy-induced nausea, take 30-60 minutes before chemotherapy.
3. What are the common side effects?
Most common side effects are headache (may be mild and self-limiting) and constipation. Less common: dizziness, drowsiness, diarrhea, or fatigue. Serious side effects include QT prolongation (irregular heartbeat) at higher doses (>16 mg) or with other QT-prolonging drugs.
4. Is Vomiplug-MD safe during pregnancy?
Ondansetron is Pregnancy Category B. It is generally considered safe for hyperemesis gravidarum (severe morning sickness) when first-line treatments have failed. However, use only under medical supervision. Some studies have reported a possible small increase in cleft palate risk.


 Call us
Call us  Contact Us
Contact Us
