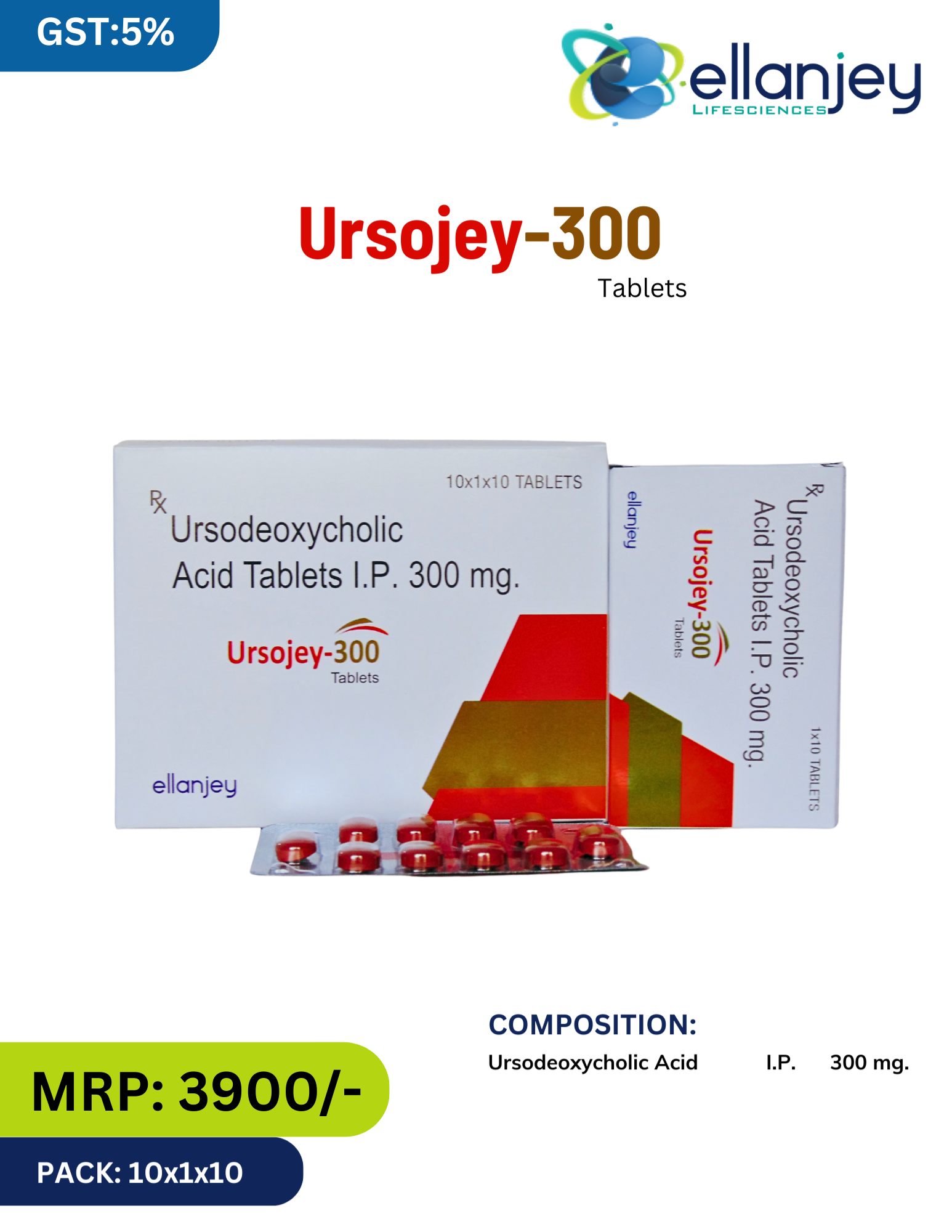
Description
Ursojey 300 by Ellanjey is a potent, hepatoprotective and choleretic medication containing Ursodeoxycholic Acid I.P. 300 mg. Ursodeoxycholic Acid (UDCA) is a naturally occurring bile acid that constitutes only a small fraction (approximately 1-3%) of the human bile acid pool. When administered therapeutically, it becomes the predominant bile acid, exerting multiple beneficial effects on the liver and biliary system.
Key Benefits:
-
First-line therapy for Primary Biliary Cholangitis (PBC) – Improves liver function, slows fibrosis progression & improves transplant-free survival
-
Dissolves cholesterol gallstones – Effective for radiolucent stones in functioning gallbladder (6-24 months therapy)
-
Gold standard for Intrahepatic Cholestasis of Pregnancy (ICP) – Reduces pruritus, lowers bile acids & improves fetal outcomes
-
Hepatoprotective – Displaces toxic hydrophobic bile acids, stabilizes cell membranes
-
Immunomodulatory – Downregulates MHC expression & reduces pro-inflammatory cytokines
FAQs:
1. What is Ursojey 300 used for?
Ursojey 300 is used for primary biliary cholangitis (PBC) – an autoimmune liver disease, dissolution of cholesterol gallstones (radiolucent, functioning gallbladder), intrahepatic cholestasis of pregnancy (ICP), cystic fibrosis liver disease, and other cholestatic liver conditions.
2. How should I take Ursojey 300 tablets?
Take exactly as prescribed – with meals to improve absorption (immediately after breakfast and dinner). Swallow whole with water. For PBC, long-term lifelong therapy is usually required. For gallstone dissolution, treatment continues for 6-24 months until stones dissolve.
3. What are the common side effects?
The most common side effect is diarrhea (mild to moderate, dose-dependent – 2-5% of patients). Other side effects include loose stools, nausea, dyspepsia, abdominal pain, headache, and dizziness. Rarely, gallstones may become calcified (discontinue therapy).
4. Is Ursojey 300 safe during pregnancy?
Yes, Ursodeoxycholic Acid is Pregnancy Category B and is actually the first-line treatment for intrahepatic cholestasis of pregnancy (ICP). It reduces itching and improves fetal outcomes. Always use under medical supervision.


 Call us
Call us  Contact Us
Contact Us
